
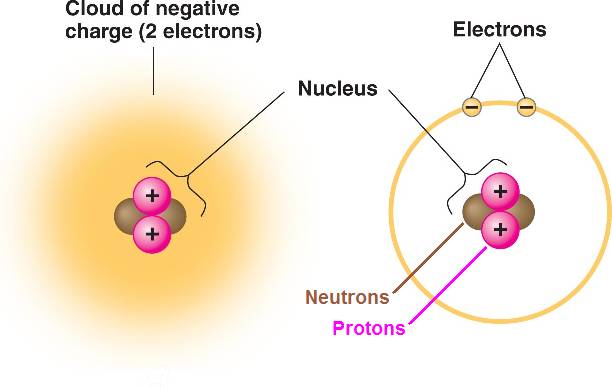
 In a helium atom,
the nucleus contains 2 neutrons which have no electrical charge,
and 2 positively charged protons.
In a helium atom,
the nucleus contains 2 neutrons which have no electrical charge,
and 2 positively charged protons.
Two electrons, which are negatively charged, move around the nucleus.
The atomic number of an element is the number of protons in the nucleus.
The mass number (atomic mass) is the sum of protons plus neutrons.